Distinct subtypes
How does LGSOC differ from HGSOC?
Initially, it was thought that LGSOC was on a continuum with high-grade serous ovarian cancer, but more recently, LGSOC has been established as a distinct subtype.3
The World Health Organization (WHO) Classification of Tumours (5th ed) defines LGSOC as an invasive serous neoplasm with low-grade malignant features.4
- LGSOCs are characterized by low mutational burden, lack of TP53 mutations, no clear germline association, and frequent MAPK-pathway alterations.5,6
Differentiating LGSOC from HGSOC
Histopathological and molecular characteristics
between LGSOC and HGSOC.
Managing LGSOC depends on distinguishing it from HGSOC.
LGSOC
HISTOPATHOLOGICAL CHARACTERISTICS:
Mild-to-moderate nuclear atypia1
Low mitotic index (12 per 10 hpf)1
molecular characteristics:
TP53 wild type7
KRAS, BRAF, NRAS, and PIK3CA mutations5,8
HGSOC
HISTOPATHOLOGICAL CHARACTERISTICS:
Tumors are pleomorphic with marked nuclear
pleomorphism (>3:1 size variation)1
High mitotic index (≥12 mitoses per 10
high-powered fields)1
molecular characteristics:
TP53 mutation and BRCA 1/2 mutations5
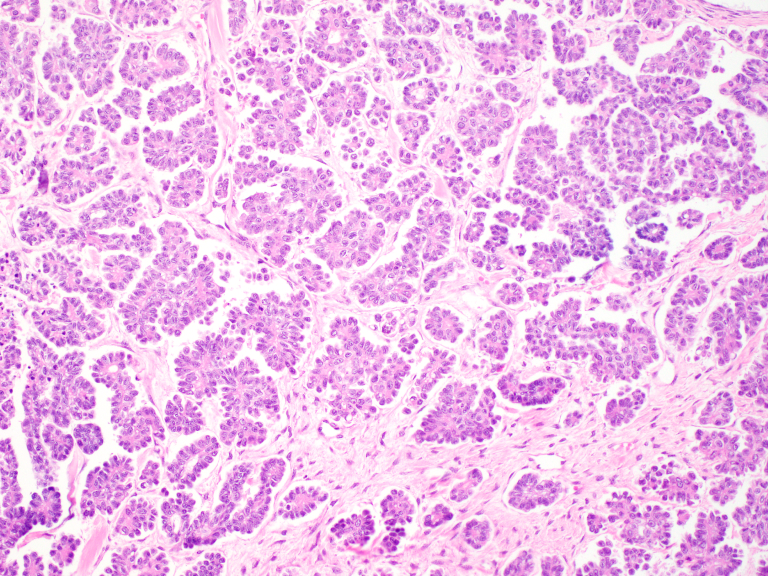
Distinguishing histological features of LGSOC and HGSOC 9
LGSOC
Low‐grade serous ovarian cancer (LGSOC) is defined by stromal invasion by tumor cells, arranged in nests and micropapillae. The nuclei are small, round, and monotonous.
HGSOC
In contrast, high‐grade serous ovarian cancer (HGSOC) shows marked nuclear pleomorphism, including tumor giant cells, and abundant mitotic figures (inset: higher magnification of boxed area with mitoses indicated by arrowheads).
Hear from experts
Low-grade vs high-grade ovarian cancer
Key characteristics of LGSOC
Germline testing
BRCA mutations rare (approximately 5%)10
Somatic testing
Can be helpful to determine prognosis and response to certain therapies11
Activating mutations in RAS/MAPK pathway promote tumorigenesis11
- KRAS mutations common (~1/3 patients)5
- NRAS5
- BRAF5
- NF18
- RAF112
Less common mutations found in LGSOC tumors: ERBB2, and PIK3CA5,13
Somatic tumor testing may identify patients for novel and emerging
treatment options or clinical trials11
~80% of patients with LGSOC are diagnosed at advanced stages, demonstrating the need for accurate diagnosis14
Healthcare provider guidelines for diagnosis

Diagnostic workup
The diagnostic workup for LGSOC typically includes clinical examination including pelvic examination, obtaining tumor markers (CA125), and imaging techniques like MRI, PET-CT, PET head to thigh, or CT which should include the chest, abdomen, and pelvis.2
NCCN Guidelines recommend both germline mutation testing and somatic tumor testing for all patients with ovarian cancer, including LGSOC.15
Imaging in LGSOC
- NCCN guidelines recommend imaging in LGSOC, including chest/abdominal/pelvic computed tomography (CT), magnetic resonance imaging (MRI), positron emission tomography (PET)-CT, or PET (skull base to midthigh), as clinically indicated.15 There is not one optimal imaging technique for low-grade serous carcinoma; however, a majority of panelists prefer CT to PET-CT.2
Histopathology Diagnosis
- LGSOC and HGSOC are distinguished using a two-tiered pathology grading system based primarily on nuclear atypia and mitotic index as a secondary feature1
Germline testing
- Germline testing for all women newly diagnosed with ovarian cancer will help to better define the prevalence of alterations in rare tumor populations such as LGSOC over time.9 Although BRCA mutations are rare in LGSOC, patients should still be offered germline testing.16
Somatic tumor testing
- At diagnosis, a somatic tumor testing panel should be conducted for LGSOC. The panel should test, at a minimum, KRAS, HRAS, NRAS, BRAF, NF1, and BRCA alterations.5,7 These tests can be helpful to determine prognosis and response to certain therapies.11
- Reasons to repeat somatic tumor testing may include unpredictable clinical behavior, clinical trial eligibility, or prolonged disease course2
- In the recurrent setting, tumor molecular analysis is recommended to include, at a minimum, tests to identify potential benefit from targeted therapeutics that have tumor-specific or tumor-agnostic action, including, but not limited to, BRCA1/2, HR status, MSI/dMMR, TMB, BRAF, FRɑ, RET, and NTRK15
- Somatic tumor testing may identify patients for novel and emerging treatment options or clinical trials.11
Pathologist consultation
- Consultation with a pathologist experienced in evaluating these lesions (LGSOC, SBT, etc.) is recommended for accurate diagnostic classification.2
Earlier diagnosis and better understanding of what drives LGSOC may help patients who may suffer for years with poor quality of life2
- Malpica A, Deavers MT, Lu K, et al. Grading ovarian serous carcinoma using a two-tier system. Am J Surg Pathol. 2004;28(4):496-504. doi:10.1097/00000478-200404000-00009
- Grisham RN, Slomovitz BM, Andrews N, et al. Low-grade serous ovarian cancer: expert consensus report on the state of the science. Published online August 17, 2023. Int J Gynecol Cancer. 2023;ijgc-2023-004610. doi:10.1136/ijgc-2023-004610
- Vang R, Shih IeM, Kurman RJ. Ovarian low-grade and high-grade serous carcinoma: pathogenesis, clinicopathologic and molecular biologic features, and diagnostic problems. Adv Anat Pathol. 2009;16(5):267-282. doi:10.1097/PAP.0b013e3181b4fffa
- WHO Classification of Tumours Editorial Board. Female genital tumours [Internet]. Lyon (France): International Agency for Research on Cancer; 2020. (WHO classification of tumours series, 5th ed.; vol. 4). https://tumourclassification.iarc.who.int/chapters/34
- Manning-Geist B, Gordhandas S, Liu YL, et al. MAPK pathway genetic alterations are associated with prolonged overall survival in low-grade serous ovarian carcinoma. Clin Cancer Res. 2022;28:4456-4465. doi:10.1158/1078-0432.CCR-21-4183
- Wong KK, Tsang YTM, Deavers MT, et al. BRAF mutation is rare in advanced-stage low-grade ovarian serous carcinomas. Am J Pathol. 2010 Oct; 177(4): 1611–1617. doi:10.2353/ajpath.2010.100212
- Sallum LF, Andrade L, Ramalho S, et al. WT1, p53 and p16 expression in the diagnosis of low- and high-grade serous ovarian carconimas and their relation to prognosis. Oncotarget, 2018; 9(22):15818-15827. doi:10.18632/oncotarget.24530
- Dey P, Nakayama K, Razia S, et al. Development of low-grade serous ovarian carcinoma from benign ovarian serous cystadenoma cells. Cancers. 2022;14(6):1506. doi:10.3390/cancers14061506
- Grisham RN, Manning‐Geist BL, Chui MH. The highs and lows of serous ovarian cancer. Cancer. 2023;1-8. doi:10.1002/cncr.34903
- Norquist BM, Harrell MI, Brady MF, et al. Inherited mutations in women with ovarian carcinoma. JAMA Oncol. 2016; 2(4):482–490. doi:10.1001/jamaoncol.2015.5495
- Grisham RN, Chui MH. Advancements in low-grade serous carcinoma of the ovary and peritoneum. Curr Oncol Rep. 2022;24(11):1549-1555. doi:10.1007/s11912-022-01315-y
- Gershenson DM. Trametinib versus standard of care in patients with recurrent low-grade serous ovarian cancer (GOG 281/LOGS): an international, randomised, open-label, multicentre, phase 2/3 trial. Lancet. 2022;399(10324):541-553. doi:10.1016/S0140-6736(21)02175-9
- Jones S, Wang TL, Kurman RJ, et al. Low-grade serous carcinomas of the ovary contain very few point mutations. J Pathol. 2012; 226(3):413–420. doi:10.1002/path.3967
- Babaier A, Mal H, Alselwi W, Ghatage P. Low-Grade Serous Carcinoma of the Ovary: The Current Status. Diagnostics (Basel). 2022;12(2):458. doi:10.3390/diagnostics12020458
- NCCN Guidelines Recommendations. Clinical Practice Guidelines in Oncology: Ovarian Cancer. Version 2.2023.
- Konstantinopoulos P, Norquist B, Lacchetti C, et al. Germline and somatic tumor testing in epithelial ovarian cancer: ASCO guideline. J Clin Oncol. 2020; 38(11):1222–1245. doi:10.1200/JCO.19.02960